Resident Program - Case of the Month
Discussion
Marginal zone lymphomas are a group of indolent non-Hodgkin lymphomas (NHL) postulated to derive from B lymphocytes in the marginal zone of secondary lymphoid tissue. It accounts for approximately 8% of all NHL cases and is the third most common B cell NHL (1,2). The average age at diagnosis is 60 years, and men and women are about equally affected (3).
Marginal zone lymphoma (MZL) includes three different entities: extranodal marginal zone lymphoma of mucosal associated lymphoid tissue (MALT), splenic marginal zone lymphoma (Splenic MZL) and nodal marginal zone lymphoma (Nodal MZL), accounting for approximately 70%, 20%, and 10% of MZLs, respectively (1,2). In the current case, the patient has splenomegaly and hepatomegaly, which may raise the consideration of Splenic MZL. However, in Splenic MZL, peripheral lymphadenopathy is extremely uncommon, and the peripheral blood commonly shows villous lymphocytes (2). The patient in the current case presents with peripheral lymphadenopathy with no circulating villous lymphocytes present, making Nodal MZL the most likely diagnosis.
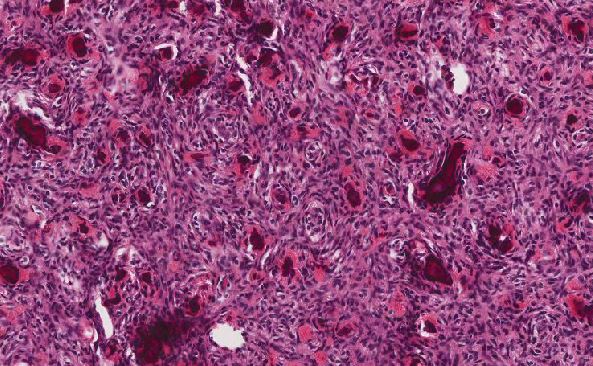
Histologically, these three subtypes of MZL share similar features (2,3). The tumor cells are small to medium sized lymphoid cells that surround reactive follicles and expand into the interfollicular areas. Follicular colonization can be present. In cases with a diffuse pattern, like in this current case, follicle remnants may be detected with immunohistochemical stains for follicular dendritic cells (e.g. CD21, CD23). The tumor cells have dispersed chromatin and abundant pale cytoplasm which are monocytoid and centrocyte-like and resemble marginal zone cells. These cells are positive for B cell markers (e.g. CD20, CD79a, PAX5, OCT2), while negative for CD3, CD5, CD10, CD23, CD30 and BCL1.
It is worth noting that large cells resembling centroblasts or immunoblasts are sometimes present, intermingled with the above described monocytoid neoplastic cells. These large cells may raise the differential diagnosis of nodular lymphocyte predominant Hodgkin lymphoma (NLPHL). However, NLPHL usually has a nodular growth pattern and contains popcorn-like large cells. The infiltrating lymphocytes in NLPHL have less cytoplasm and are not monocytoid.
Monoclonal plasma cells with Dutcher bodies may be prominent in MZL (2,3). When present, they may pose a problem for differential diagnosis from lymphoplasmacytic lymphoma or nodal plasmacytoma. The presence of remnants of follicular dendritic cell meshworks is suggestive of follicle colonization and favors the diagnosis of Nodal MZL (2).
The B- and T-cell gene rearrangement analysis by PCR was performed in the current case. The results showed that the clonal immunoglobulin (Ig) kappa gene rearrangement was present while T cell gamma chain and beta chain gene rearrangements were not detected, which supports the diagnosis of a B cell neoplasm. Indeed, in Nodal MZL the Ig genes are clonally rearranged with predominant mutations in IgHV3 and IgHV4 (2). Nodal MZL often shows gains of chromosomes 3 and 18 and loss of 6q23-24, a similar finding seen in MALT and Splenic MZL. However, the recurrent translocations (e.g. t(11;18), t(14;18)) associated with extranodal MZL are not detected in Nodal MZL or Splenic MZL (2).
MZLs represent a clinical challenge, not only because of the diagnostic pitfalls, but also because their relative rarity, especially of individual subtypes; because of this, a standard management strategy has not been adopted (4). The three subtypes of MZL differ in prognosis and outcomes. Therefore, identification of subtype and clinical characteristics is important to determine the treatment plan (1,2). It has been reported that prognosis in patients with MALT is statistically significantly better than that in patients with Splenic MZL or Nodal MZL. The 5-year survival rate was estimated to be 88.7% for MALT, compared with 79.7% for Splenic MZL and 76.5% for Nodal MZL (5).
References:
- Nakamura S, Ponzoni M.Pathology. Marginal zone B-cell lymphoma: lessons from Western and Eastern diagnostic approaches. 2020 Jan;52(1):15-29
- Swerdlow SH, Campo E, Harris ES et al. WHO classification of tumors of hematopoietic and lymphoid tissues. Revised 4th 2017
- Reid R, Friedberg JW. Management of marginal zone lymphoma. Oncology (Williston Park) 2013;27:840, 842, 844
- Rosand CB, Valla K, Flowers CR, Koff JL. Effective management strategies for patients with marginal zone lymphoma. Future Oncol. 2018 May;14(12):1213-1222
- Olszewski AJ, Castillo JJ. Survival of patients with marginal zone lymphoma. Cancer. 2013;629-38.

 Meet our Residency Program Director
Meet our Residency Program Director