Resident Program - Case of the Month
September 2018 - Presented by Trevor Starnes, M.D.
Discussion
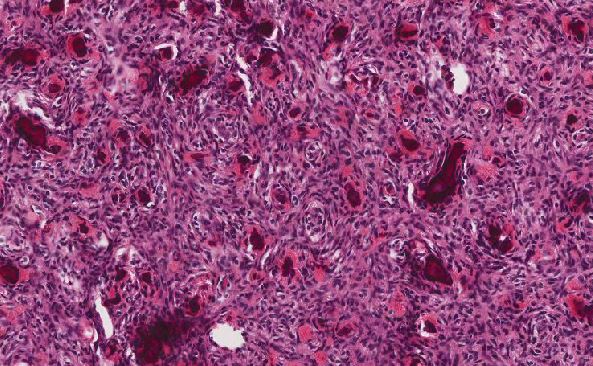
The histology of this pancreatic mass reveals infiltrating sheets of large cells with an elevated nuclear to cytoplasm ratio, open chromatin, and prominent nucleoli. This is morphologically compatible with immature myeloblasts. The native pancreatic tissue is effaced. The most pertinent immunohistochemical stains showed that the malignant cells are myeloperoxidate (MPO) positive, and faintly positive for CD45. MPO is a specific stain for myeloid processes, and CD45 or leukocyte specific antigen (LCA) is faint in immature myeloid cells and has increasing positivity as they mature. Additionally, CD43 IHC was positive, which is expressed by T-cells, but also expressed in immature hematopoietic cells. For monoblastic variants, MPO may be negative but positivity for CD68 is sensitive.
Myeloid sarcoma is related to acute myeloid leukemia (AML) in that it also consists of immature myeloid blasts, but myeloid sarcoma is an extramedullary tumor. It has gone by several names since its discovery in 1811. Early on, it was called a chloroma, since it tends to have a green color from myeloperoxidase. More recently, it has also been known as granulocytic sarcoma.
Myeloid sarcoma can occur on its own, but may be concurrent with AML, myelodysplastic syndrome, chronic myeloid leukemia, or myeloproliferative disorders. Furthermore, most patients with isolated myeloid sarcoma later develop AML after months or even years. Though considered a rare diagnosis, myeloid sarcoma is found in about 8% of autopsies on patients who died with AML. For isolated myeloid sarcoma, history of chemotherapy is a risk factor.
De novo myeloid sarcoma (i.e. without a concurrent myeloid neoplasm involving the marrow) constitutes about 27% of myeloid sarcomas in adults, and is very rare in the pediatric population. Again, the pancreas is an unusual site. More common sites of involvement include lymph nodes, skin, and soft tissues. In children in particular, the skin and orbit are the most common sites. However, it is believed that myeloid sarcoma can occur at almost any site and age.
Myeloid sarcoma is treated similarly to AML with chemotherapy and bone marrow transplant, even prior to bone marrow involvement. Timely diagnosis and initiation of AML-targeted therapy appears to be key to survival. Prognosis seems to also be affected by site of involvement, e.g. cutaneous disease is more aggressive than orbital. Pancreatic disease is rare and so prognosis is speculative. Some of these patients died due to recurrence of AML/myeloid sarcoma.
Molecularly, different sites of involvement tend to have different translocations. Intraabdominal involvement is associated with inv(16), while orbital involvement is associated with t(8;21), and trisomy 8 is associated with cutaneous disease.
Differential diagnosis
*Myeloid sarcoma is most often confused with large cells lymphomas, which can be indistinguishable on H&E. Approximately 46% of myeloid sarcomas have been initially misdiagnosed as a large cell lymphoma historically. Immunohistochemical staining is useful for making the distinction (MPO being specific, but again can be absent in monoblastic cases), but myeloid sarcoma can be positive for T and B cell markers, even CD3 and CD79a, respectively.
*Pancreatoblastoma, the most common pancreatic malignancy of childhood, was in the radiological differential. However, microscopically it is characterized by epithelial cells with acinar endocrine, or ductal differentiation and squamoid corpuscles, none of which is seen here.
*Myeloid metaplasia, a form of extramedullary hematopoiesis, more often involves an enlarged spleen or liver, would not efface the native architecture, and would be expected to show a spectrum of maturation. The bone marrow also would not be unremarkable, e.g. myeloproliferative or myelofibrosis.
*Undifferentiated carcinoma of the pancreas can have a variety of appearances including spindled cells, anaplastic giant cells, or carcinosarcoma, and so some cases could resemble myeloid sarcoma on H&E. While undifferentiated carcinomas can be negative for keratin, positivity for MPO in this case confirms myeloid sarcoma.
References
Hagen PA, Singh C, Hart M, Blaes AH. Differential Diagnosis of Isolated Myeloid Sarcoma: A Case Report and Review of the Literature. Hematology Reports. 2015;7(2):5709. doi:10.4081/hr.2015.5709.
Kudva R, Monappa V, Salanke G, Valiathan M, Anuradha CK, Geetha V. Myeloid sarcoma: A clinicopathological study with emphasis on diagnostic difficulties. Journal of Cancer Research and Therapeutics. 2017;13(6):989-993.
Messager M, Amielh D, Chevallier C, Mariette C. Isolated granulocytic sarcoma of the pancreas: A tricky diagnostic for primary pancreatic extramedullary acute myeloid leukemia. World Journal of Surgical Oncology. 2012;10(13).
Neiman RS, Barcos M, Berard C, Bonner H, Mann R, Rydell RE, Bennett JM. Granulocytic sarcoma: a clinicopathologic study of 61 biopsied cases. Cancer. 1981 Sep 15;48(6):1426-37.
Pileri SA, Orazi A, Falini B. (2017) Myeloid sarcoma. In SH Swerdlow, E Campo, NL Harris, et al. (eds), WHO Classification of Tumours of Haematopoietic and Lymphoid Tissues, Revised 4th Edition. pp. 167-168.
Samborska M, Derwich K, Skalska-Sadowska J, Kurzawa P, Wachowiak J. Myeloid sarcoma in children – diagnostic and therapeutic difficulties. Contemporary Oncology. 2016;20(6):444-448. doi:10.5114/wo.2016.65602.
Sternberg, S. S., Mills, S. E., & Carter, D. (2010). Sternberg's diagnostic surgical pathology (5th ed.). Philadelphia: Wolters Kluwer Health/Lippincott Williams & Wilkins.

 Meet our Residency Program Director
Meet our Residency Program Director