Case of the Month
July 2017 - Presented by Dr. Ruijun Su (Mentored by Dr. Eric Huang)
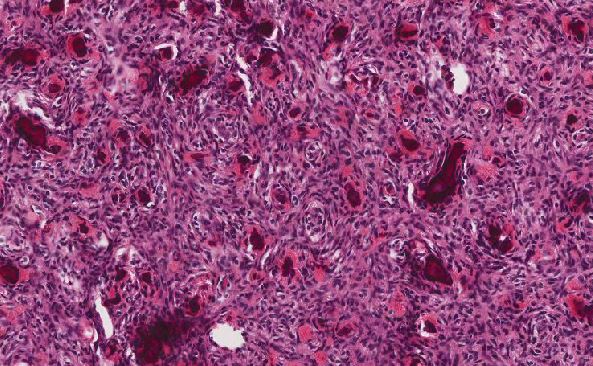
Discussion
The 2008 World Health Organization scheme defined distinct categories of B-ALL by immunophenotyping with subtypes substantially amplified by cytogenetics and molecular diagnostic tools. Other variants of B-ALL may include cytoplasmic granules, aplastic presentation, eosinophilia, relapse of lymphoblastic leukemia and secondary ALL [1]. In the current case of CNS relapse of B-ALL, the morphological differential diagnosis includes the following:
-
"B lymphoblast crisis" of CML with CNS relapse. Although rare, this is an important clinical scenario to consider, as the initial CML may be indolent and undiagnosed. When blast crisis occurs, myeloblasts are more common, consisting 70% of the cases. However, 20% can present as B lymphoblasts, with occasional cases of T lymphoblastic, monoblastic, megakaryoblastic, erythroblastic and multilineage blastic crises [2, 3], all carrying worse prognosis. Pattnaik et al. reported a case of t(9;22) CML presented as blast crisis mimicking ALL in a child. The morphologic presence of basophils and basophilic myelocytes can be seen in peripheral blood and lymph node aspirate smears; however, pre-B cell lineage by immunomarkers in flow cytometry played an important role in diagnosing the case as CML with lymphoblastic crisis [4]. At the molecular level, the most common mutations detectable at the loci of cyclin-dependent kinase inhibitor 2A/2B (CDKN2A/B) (50% of cases) and Ikaros transcription factor (IKZF1) (55% of cases) can be seen in lymphoid transformation of CML [5–7]. Homozygousdeletion of p16 tumor suppressor gene is also associated with lymphoid transformation of CML [8].
-
Biphenotypic acute leukemia. In cases with dimorphic blast population, the possibility of a mixed phenotype acute leukemia with t(9;22) (q34; q11.2) should be considered. This is a very rare form of leukemia (<1%) and patients usually presented with high white blood cell count, similar to those with Ph+ ALL. Our patient did not have a mixed phenotype on flow cytometric analysis.
-
“Cup-like” leukemic cells in AML. To some extent, the leukemia cells in our case resemble cup-like leukemic cells in AML. The cup-like nuclear phenotype was defined by the presence of nuclear invaginations in~25% of the nuclear diameter. Myeloperoxidase activity can be observed in a distinctive granular pattern within the areas of nuclear invagination and electron microscopy revealed an accumulation of mitochondria in the nuclear indentation. This morphology is associated with high blast counts in the peripheral blood and bone marrow, especially in AML M1 (former FAB classification). Gene mutation of both NPM1 and FLT3-ITD or TKD strongly correlated with the cup-like nuclear morphology. Although it is well recognized that blasts with cuplike nuclei are associated with AML, including AML with t(9;22)(q34;q11.2), Hu et al. reported that they can be encountered in B-ALL [9]. The morphology of clefted-nuclei looking monoblasts and promonocytes overlaps with that of cup-like nucleated cells. Therefore, AMLs (M4 and M5) easily enters the differential diagnosis as these entities often associated with CNS involvement.
- Burkitt leukemia, or L3-ALL (former FAB classification). Typical L3 blasts are medium to large in size and homogeneous. The nuclei are regular and round to oval in shape. One or more prominent nucleoli are present. The amount of the cytoplasm is moderate and contains prominent vacuoles. Cytochemistry of L3 blasts is always negative for MPO and NSE, while intensely positive for cytoplasmic methyl green pyronine; cytoplasmic vacuoles stain strongly with Oil red O [10]. All L3 leukemias are surface immunoglobulin (sIg) positive and are of B cell lineage. A leukemic phase can be seen in patients with bulky disease of Burkitt lymphoma,
but rare cases can be present purely as acute leukemia with peripheral blood and bone marrow involvement [1]. CNS involvement is common in L3 ALL, thus influencing both treatment and prognosis. Recognizing this entity is important as patients with L3 morphology showed improved outcomes when treated with specific algorithms [11, 12]. Some studies found that L3 leukemia can be cured with aggressive, rapidly cycling lymphoma-like chemotherapy regimens [11, 13, 14]. L3 ALL is associated with a variety of translocations including the c-MYC protooncogene to the immunoglobulin gene locus t(2;8), t(8;12), and t(8;22). In fact, translocations involving chromosome 8 (MYC gene) are present in 100% of B-ALL with L3/Burkitt morphology and clonal mature sIg [11]. In our patient, cytogenetic studies showed a very complex karyotype with multiple chromosome changes, most critical of which is the t(9;22)(q34.1;q11.2). Another important translocation is t(8;9) that contains alterations in chromosome 8. We speculate that such combination of unusual genetic abnormalities may have led to the unique morphology of “AML-like” blasts in our patient. In addition, other genetic abnormalities such as deletion of 9p are also an adverse risk factor for B-ALL [15]. Additional copies of chromosome 4 are also present with unclear clinical significance. All of these genetic abnormalities may have contributed to the aggressive clinical behavior and rapid demise in this patient. The co-existence of t(9;22)(q34;q11) and t(8;9)(q13;p13) is rare. Some studies have suggested that the occurrence of a second translocation involving chromosome 8 in addition to BCR-ABL1 is strongly associated with advanced stage of disease, as an additional growth advantage of leukemic cells is conferred [16]. Our patient may have experienced blast crisis/relapse due to harboring both clones when both translocations are present.
There is growing consensus that identifying factors that increase risk of leukemic CNS involvement can lead to early modification of protocols aiming at treatment and CNS prophylaxis. It is well established that leukemic patients can benefit from concurrent cytology and flow cytometry studies, as patients with any positive result from either study may receive early intervention, even when they are clinically asymptomatic.
In summary, blasts in CNS relapsed B-ALL with t(9;22)(q34;q11) BCR/ABL1 can mimic myeloblasts in AML. The aforementioned differential diagnoses should be considered when encountering challenging scenarios similar to our case. The combination of morphologic, immunophenotypic and cytogenetic/molecular studies is the best approach to render an accurate diagnosis.
References
- Swerdlow SH, Campo E, Harris NL, et al. WHO Classification of Tumors of Haematopoietic and Lymphoid Tissues. Lyon, France: IARC Press; 2008.
- Tsieh Sun. Flow Cytometry, Immunohistochemistry, and Molecular Genetics for Hematologic Neoplasms. 2nd ed. Wolters Kluwer/Lippincott Williams & Wilkins 2008.
- Atallah E, Talpaz M, O’brien S, et al. Chronic myelogenous leukemia in T cell lymphoid blastic
phase achieving durable complete cytogenetic and molecular remission with imatinib mesylate (STI571;
Gleevec) therapy. Cancer. 2002;94(11):2996-2999. - Patna K, Kar A, Palai S, et al. Philadelphia positive chronic myeloid leukemia mimicking acute lymphoblastic leukemia in a child. African Journal of Medical and Health Sciences. 2013; 12 (2): 116-119.
- Mullighan CG, Williams RT, Downing JR, et al. Failure of CDKN2A/B (INK4A/B-ARF)-mediated tumor
suppression and resistance to targeted therapy in acute lymphoblastic leukemia induced by BCR-ABL.
Genes Dev. 2008;22(11):1411-1415. - Mullighan CG, Miller CB, Radtke I, et al. BCR-ABL1 lymphoblastic leukaemia is characterized by the deletion of Ikaros. Nature. 2008;453(7191):110-114.
- Nakayama H, Ishimaru F, Avitahl N, et al. Decreases in Ikaros activity correlate with blast crisis in patients with chronic myelogenous leukemia. Cancer Res. 1999;59(16):3931-3934.
- Sill H, Goldman JM, Cross NC, et al. Homozygous deletions of the p16 tumor-suppressor gene are associated with lymphoid transformation of chronic myeloid leukemia. Blood. 1995;85(8):2013-2016.
- Wang W, Xie W and Hu S. Cup-like blasts in Blymphoblastic leukemia. British Journal of Haematology,
2015, 170, 596. - Chiaretti S, Zini G and Bassan R. Diagnosis and subclassification of Acute Lymphoblastic Leukemia.
Mediterr J Hematol Infect Dis 2014; 6: Open Journal System. - Lee EJ, Petroni GR, Schiffer CA, et al. Brief-duration high-intensity chemotherapy for patients with small noncleaved-cell lymphoma or FAB L3 acute lymphocytic leukemia: results of cancer and leukemia group B study 9251. J Clin Oncol 2001;19 (20): 4014-4022.
- Hoelzer D, Ludwig WD, Thiel E, et al. Improved outcome in adult B-cell acute lymphoblastic leukemia.
Blood 1996;87 (2): 495-508. - Fenaux P, Lai JL, Miaux O, et al. Burkitt cell acute leukaemia (L3 ALL) in adults: a report of 18 cases. Br J Haematol 1989;71 (3): 371-376.
- Reiter A, Schrappe M, Ludwig WD, et al. Favorable outcome of B-cell acute lymphoblastic leukemia inchildhood: a report of three consecutive studies of the BFM group. Blood 1992; 80 (10): 2471-2478. 19.
- Nahi H, Hägglund H, Ahlgren T et al. An Investigation Into Whether Deletions In 9p Reflect Prognosis In Adult Precursor B-Cell Acute Lymphoblastic Leukemia: A Multi-Center Study Of 381 Patients.Haematologica. 2008; 93: 1734-1738.
- Solari L, Bauer T, Dicker F, et al. A novel recurrent AML1-ETO fusion: tight in vivo association with BCR-ABL1. Leukemia 2013; 27, 1397-1400.

 Meet our Residency Program Director
Meet our Residency Program Director